|
11/7/2023 0 Comments Dynamic viscosity mu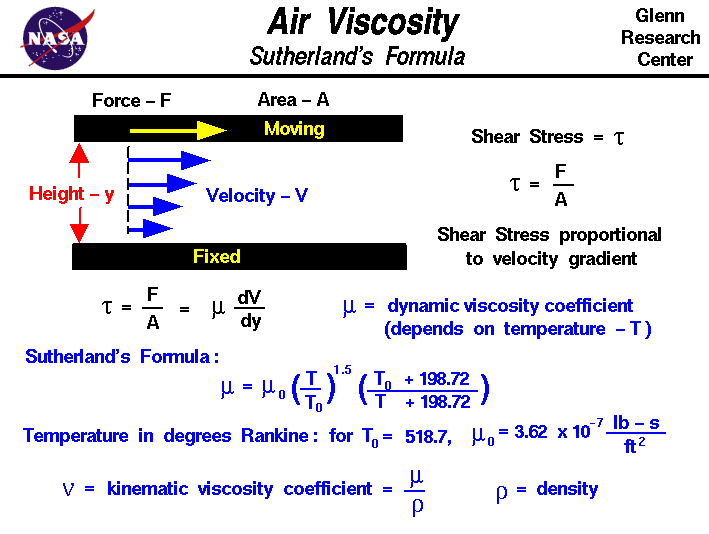
This seems to be a mistake that no-one resolved in public forums. So I guess someone made a mistake here and everybody just re-used the same. nnNevertheless, as we already take the effecitve viscosity (which normally comes from the turbulence model), we should not include the first term and also there should be no need to divide the second part by the turbulent Prandtl number as is should be done in the turbulence class already. However, the kinematic viscosity is around 1.8 e-5. However, it seems that it should be related to the molecular viscosity of air which is:density * kinematic viscosity. The pre-term C_RHO * 2.88e-5 does not make any sense to me. In the equation above we clearly find the effective viscosity (C_MU_EFF). Therefore, in the diffusion equation we should have either:mu + mu_t or mu_eff. As we are using the Boussinesq Hypothesis + Reynolds Averaging method (bla blub), we end up with different turbulence models which at the end will just do one thing. However, the diffusion of the air is basically the molecular viscosity of the air (at the cell temperature) plus the increase due t o turbulence. Hence, we do need both terms in our conservative equations. First of all, the air can exchange via advection and due to molecular transport phenomona. From my personal perspective, the equation given there is wrong. I do have some questions regarding the AoA calculation mentioned above.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
